Louisville’s PharMerica is still a defendant in federal cases in which big drug makers have paid billions in fines
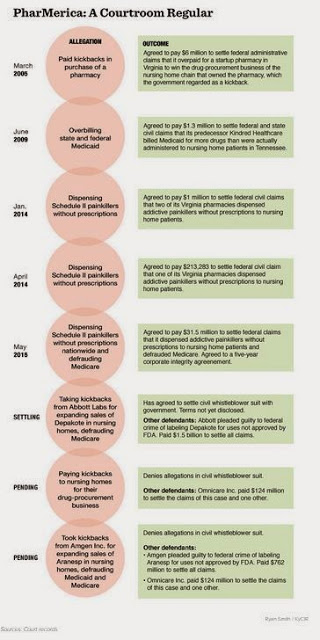
PharMerica manages drug benefits for nursing homes, hospitals and assisted living facilities. McNair paints a dismal picture of nursing homes and says they are ripe for this type of abuse, writing that they house “people with age-weakened bodies, multiple ailments and, often, severe mental impairment. Many are over-medicated. Many have no visitors. A third of them will die within a year of admission.” (Click on chart for larger version)
 |
McNair notes that a whistleblower lawsuit first called attention to Abbott Labs, which pled guilty in 2012 to a criminal charge, settled civil kickback and fraud claims, and paid $1.5 billion in fines for its role in paying millions of dollars in “rebates” to get pharmacy companies to increase prescriptions for an anti-seizure drug, Depakote, for uses beyond its Food and Drug Administration approval. Medicaid payments for this drug “went on to top $7 billion,” McNair reports.
Amgen also enlisted these same pharmacy companies to promote its anemia drug, Aranesp, for uses beyond its FDA approval, and after pleading guilty settled civil kickback and fraud charges and paid a total of $762 million in fines.
These two cases brought more government attention to such schemes, which are “standard practice in the pharmaceutical industry,” and also on the pharmacy companies that are on the receiving end of the payoffs, McNair writes.
McNair describes PharMerica as the “second-biggest operator of nursing home pharmacies in the country” and writes that it had ” $1.9 billion in revenue last year,” making it the “10th-biggest publicly traded company in Kentucky, according to rankings by The Lane Report.” Since 2007, the chief executive has been Gregory Weishar (pronounced WISH-er) .
The Abbott Labs and Amgen lawsuits assert that PharMerica gave “certain drugs to nursing home patients in return for drug company kickbacks, not because they were the “right medication.””
McNair reports that the suits were filed by drug company insiders who have knowledge of these payoffs disguised as “rebates” or “discounts.”
“PharMerica denies the claims,” writes McNair. But the company has been in this type of case many times since 2005, McNair reports: It has agreed to pay $40 million in fines to settle federal complaints, five additional closed cases connected to this company.
McNair also reports that just last week, the Justice Department said PharMerica will pay $31.5 million for dispensing addictive painkillers to nursing home patients without prescriptions, then falsely billing Medicare. As part of this settlement, PharMerica also agreed to a five year “corporate integrity agreement,” which McNair notes later in the article are rarely enforced.
McNair goes on to list the details of several other cases PharMerica has been involved in, one of them “deemed so flagrant that the inspector general sought to ban PharMerica from federal health-care programs for 10 years.”
PharMerica declined to make its executives available for an interview with the Kentucky Center for Investigative Reporting but said in a statement: “PharMerica is committed to outstanding compliance and the highest standards of ethical conduct, and we are diligent in ensuring that we comply with all applicable law and regulation,”
Jan Scherrer, vice president of Kentuckians for Nursing Home Reform, a non-profit advocacy group based in Lexington, told McNair that the CEOs of companies involved in kickback schemes should be held personally accountable, “These are not victimless crimes,” he said.
“It’s the same players — PharMerica and Omnicare,” Scherrer continued. “They keep doing this over and over and over, and all they get is a fine. And for them that fine is nothing more than the cost of doing business.” (Read more of this detailed report by clicking here.)
Excellent piece by Jim McNair. I'd like to suggest adding 'nursing homes' to your labels list.