Opinion: Covid-19 vaccine effectiveness declines rapidly; seniors need to keep up their immunity, and we need a better vaccine
By Kevin Kavanagh for Infection Control Today

Reuters reported that the committee concluded, “There is not sufficient evidence to recommend more than 1 Covid-19 booster shot a year for older people and those with weakened immune systems,” but the committee did voice some flexibility. It needs to be stressed that there was an absence or lack of data—rather than a presence of data—indicating the durability of vaccine immunity, justifying annual boosters.
There was no vote regarding the timing of booster doses, but there was nowhere near a unanimous consensus. Michael Hogue of the American Pharmacists Association said, “We want those clinicians to be able to make good decisions for the individual patient based upon their comfort and desire as long as we have safety in mind, and it is clear that we do have a very safe vaccine with our bivalent vaccine. So, I feel that flexibility needs to be put into this some way, with both older adults and people with immunocompromising conditions.”
I did not hear calls for approving a more frequent administration schedule. One committee member said those over 65 should be allowed to discuss off-label use with their physician in order to receive the booster sooner. Almost all the discussion centered on messenger-RNA vaccines, with little mention of Novavax or the urgent need for newer, more durable vaccines.
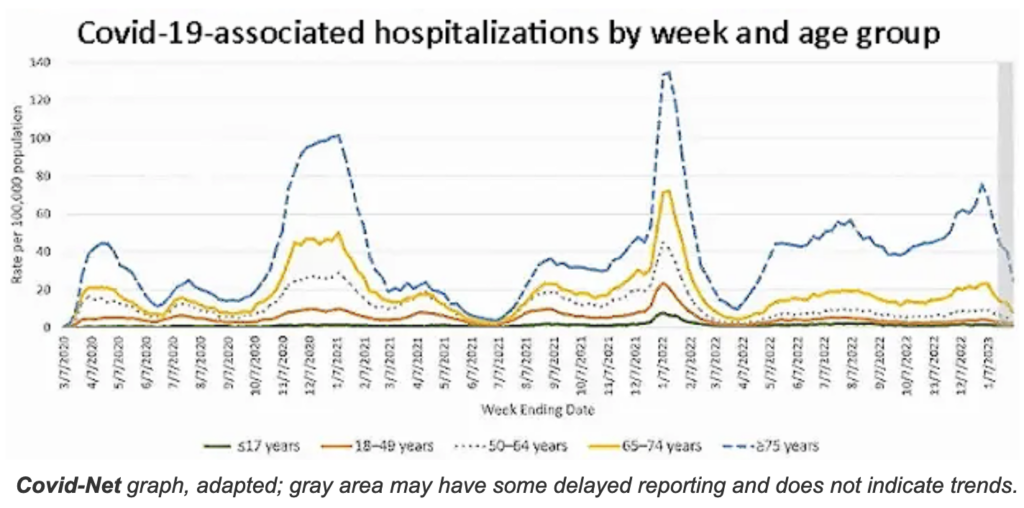
Vaccine effectiveness is an important point. Although much of the younger population received a get-out-of-hospital free card for the latest variant, senior citizens were in its crosshairs. Since May 2022, those over 75 had a higher rate of hospitalization than in the Delta surge; those 65 to 75 had about the same. Both age groups continue to be at high risk for death and disability.
However, when I saw data on effectiveness of the monovalent vaccines, the two-dose-or-more immunization that preceded the bivalent vaccine, I felt foreboding. The data showed that for those 65 years and older, the effectiveness of two or more monovalent doses at preventing hospitalizations fell to 28% in less than a year.
For the bivalent booster, effectiveness against hospitalization fell rapidly, from 52% at a median of 32 days to 31% at a median of 74 days (67-85 days) after the last dose. It should be noted that this effectiveness is on top of some residual immunity from the monovalent vaccine, since the monovalent vaccine was used as the reference.
It is critical for the elderly to keep their immunity as high as possible. Monoclonal antibodies are no longer effective with the new variants, and far too many cannot receive Paxlovid because of drug interactions. Molnupiravir is often not prescribed since it works by creating viral mutations and has been implicated in speeding variant evolution.
At the conclusion of the CDC committee meeting, my primary impression was that we senior citizens might be viewed as expendable. Far too few policymakers are concerned about our well-being and willing to make the hard decisions that must be made to assure our safety during this pandemic. After looking at the data, I will consult my physician about receiving a booster on an accelerated schedule, possibly at six months.
Kevin Kavanagh is a retired physician from Somerset and chairman of Health Watch USA. This is an edited version of his original article, which was first published in Infection Control Today.